Sheep and goat fecal analysis is an important component for goat farmers with worm and Coccidia infestations. Animal breeders including horses, rabbits, cats, dogs and cattle may also have these same issues. Worm and Coccidia parasites alone kill more goats than all other illnesses combined. A regular established program of microscopic examination of goat "pills" (feces) can help farmers find and treat parasites before they kill many of the animals.
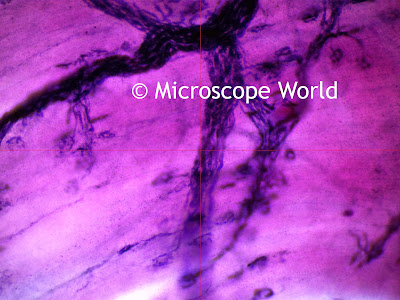
Coccidia is a protozoan parasite. This protozoan causes Coccidiosis, which can be one of the most devastating diseases to livestock, particularly among young kids. Coccidiosis causes watery diarrhea that is sometimes bloody and can be life-threatening to young animals. The presence of Coccidia in the intestines does not mean the animal is actually suffering from Coccidiosis. The protozoans only cause disease when their numbers become so great that damage is caused to the host. The image above is Coccidia during the infective stage. The oocyst contains two sporocysts.
 |
| Coccidia from a cat fecal sample. Photo: Joel Mills |
This is a list of some species of Coccidia found in animals:
- Eimeria - found in rabbits, poultry, sheep, horses, swine and goats
- Toxoplasma - found only in cats
- Isospora - found in dogs, cats, primates and swine
- Neospora Canium - found in dogs
- Sarcocystis - found in carnivores
Materials required to test for Coccidia Infestations include:
- Simple biological microscope (400x magnification needed)
- Blank glass slides
- Cover slips
- Cheesecloth
- Test Tubes
- Stir Stick (pick one up next time you get coffee)
- Fecal floatation solution (you can use sugar or salt)
- Test tube holding rack (you can use a styrofoam block with holes punched in it)
Fecal Analysis Procedure:
Mix up the flotation solution (it should be saturated, so you want to dissolve as much solid in the water as it will hold). Saturated salt solution requires a pound of salt in a little under 5 cups of water. Collect fresh feces using a small jar for each animal. Be sure to label with the date, time and animal. Place 3-4 goat pellets into a test tube and pour in just enough flotation solution to cover them. Stir and mix well with the stir stick. Strain with the cheese cloth to remove large particles and pour the strained liquid into a clean test tube. Fill up the test tube with more liquid so it reaches the very top. Place a cover slip over the top of the test tube and wait 30 minutes. Over time the eggs will float up to the top and adhere to the glass plate.
Carefully remove the cover slip and lower it at an angle over a microscope slide with the sample between both pieces of glass. Examine the specimen for worm eggs and Coccidia Oocysts. Start with the lowest magnification (40x) on the microscope and move up to 400x and if you have it, 1000x.
You should be able to identify Coccidia Oocysts, Nematode Eggs and Taperworm eggs. When performing a fecal count a general guide is that a level of about 500 eggs per gram of feces would indicate a next step of worming. If a regular testing system is in place (every 4-8 weeks is ideal) the best indication is when there is a dramatic increase in egg counts.